Multiplexed rapid technologies for sexually transmitted infections: a systematic review - The Lancet Microbe

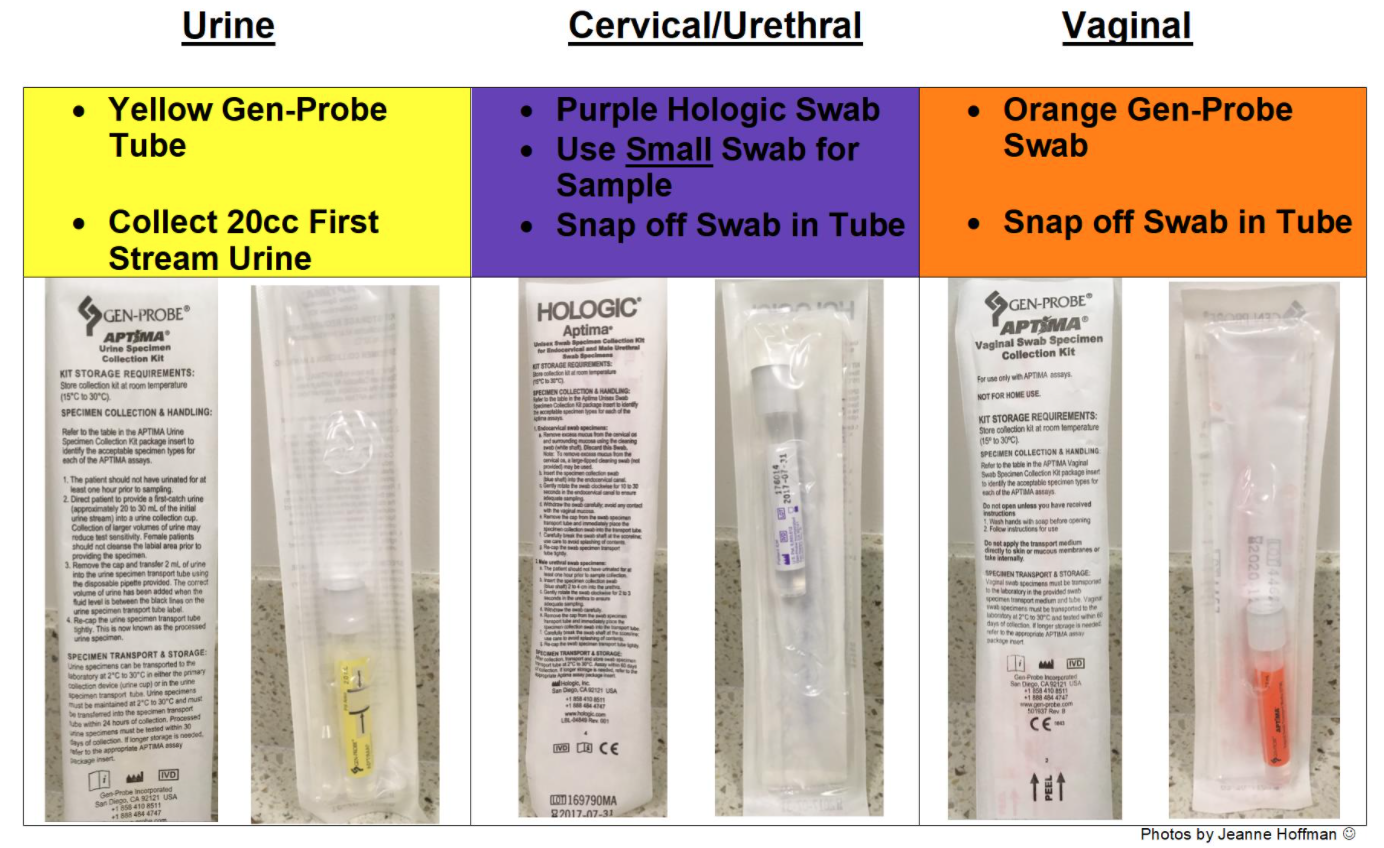
Chlamydia trachomatis/Neisseria gonorrhoeae (CT/NG) – Nucleic Acid Amplification Testing (NAAT) | Public Health Ontario

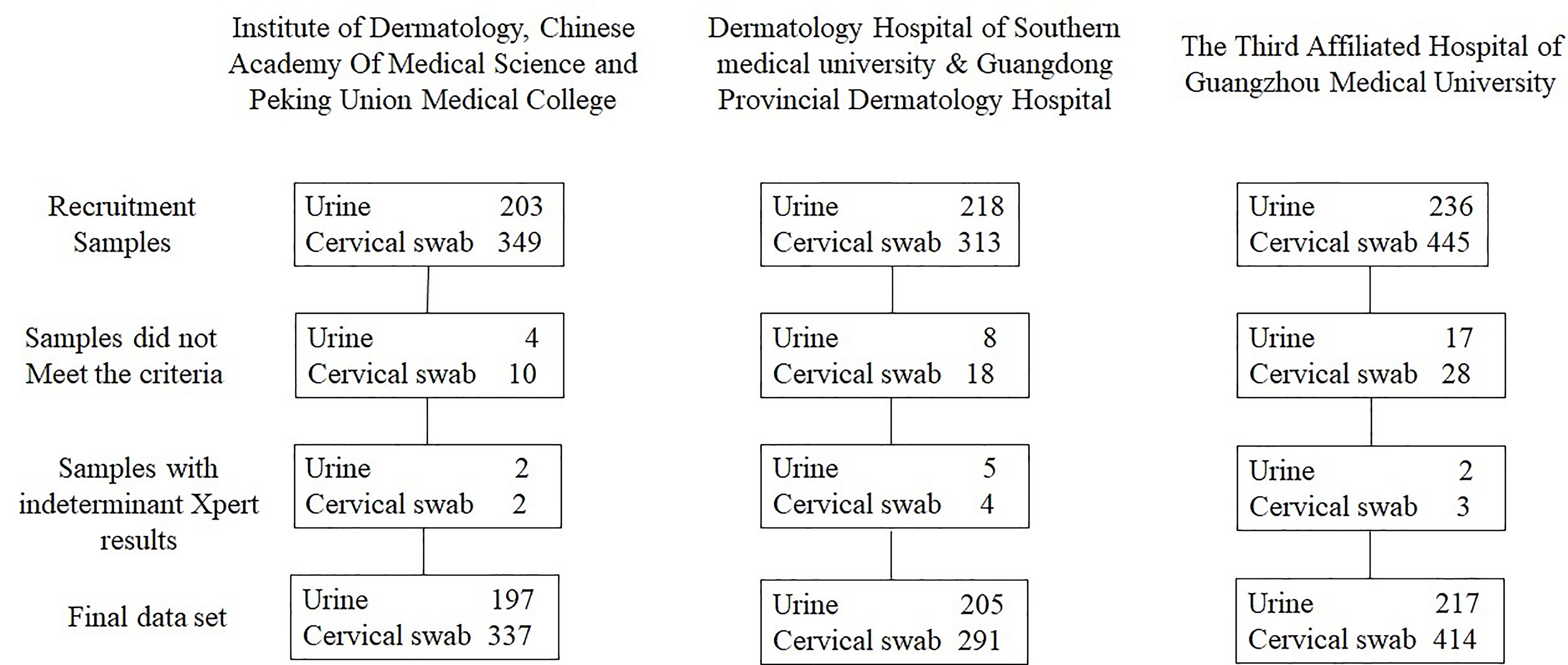
Performance of a single-use, rapid, point-of-care PCR device for the detection of Neisseria gonorrhoeae, Chlamydia trachomatis, and Trichomonas vaginalis: a cross-sectional study - The Lancet Infectious Diseases
Service Update: Chlamydia trachomatis and Neisseria gonorrhea – Nucleic Acid Amplification Testing – Updated Process for Sam