Sanofi and GSK receive up to $2.1B from the U.S. to deliver COVID-19 vaccine | 2020-07-31 | BioWorld

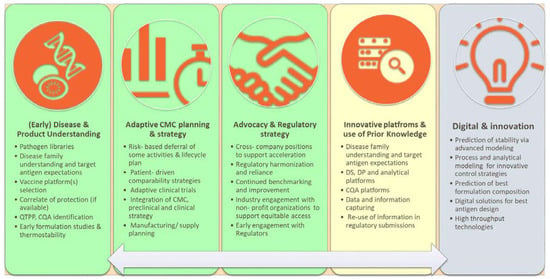
Vaccines | Free Full-Text | CMC Strategies and Advanced Technologies for Vaccine Development to Boost Acceleration and Pandemic Preparedness

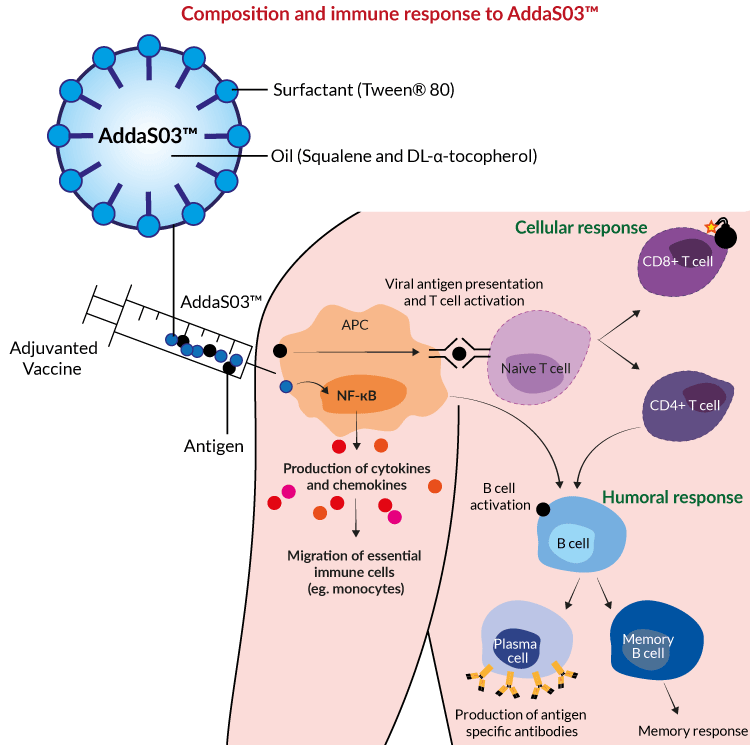
Vaccines | Free Full-Text | Use of Stability Modeling to Support Accelerated Vaccine Development and Supply

CureVac and GSK announce promising preclinical data for next-gen COVID-19 vaccine - Drug Discovery and Development
Cervarix™, the GSK HPV-16/HPV-18 AS04-adjuvanted cervical cancer vaccine, demonstrates stability upon long-term storage and under simulated cold chain break conditions: Human Vaccines: Vol 5, No 7
Statement on Rotarix® and Vaccine Vial Monitor (VVM) compliance Rotarix® is produced by GlaxoSmithKline Biologicals SA (here b